Where Are We With Organoid Cryopreservation?
You can’t perfuse an organoid with CPAs like an organ, but you can’t just dunk it in CPA like a cell suspension either. So how do you freeze a 3D tissue that sits exactly between the two modalities?
Organoids, the self-organizing 3D tissues, now underpin disease modeling, drug screening, and early regenerative medicine concepts. We need a lot of them for science, and usually they are grown on demand. But is there a point preserving the entire organoid, when we can just regrow a new one from cryopreserved cells relatively easily? Surprisingly, yes. Cryopreserving whole organoids has significant value over simply regrowing them from cells, as it preserves complex 3D structures, cell interactions, and functional maturity that dissociation often disrupts, saving weeks of culture time and reducing variability for reproducible research. Once an organoid is dissociated, the researchers lose its architecture, its maturity, its gradients, its disease phenotype, and the exact biological state it reached over weeks or months. Regrowing produces a new organoid, not the same organoid. For drug testing, patient-derived samples, developmental studies, and any work that relies on structural integrity or temporal identity, whole-organoid preservation is the only way to freeze a mature system in time and revive it exactly as it was. But can we even do it?
Most cell cryopreservation is routine. Whole-organ vitrification has taken major steps forward in rats and even toward liter-scale CPA volumes (I talk about it in the last post). Organoids sit in an awkward middle ground: too large and structured to behave like simple cell suspensions, too small and avascular to benefit from classic organ perfusion strategies. So where are we really, in 2025–2026, with organoid cryopreservation?
What has actually been cryopreserved and functionally revived?
Intestinal organoids ✅
Human intestinal and colonic organoids can be frozen, thawed, and expanded with preserved stemness and the ability to regenerate epithelium-only enteroids/colonoids. Detailed protocols describe dissociating organoids from matrix, resuspending them in DMSO-containing freezing media, long-term storage in liquid nitrogen, and subsequent thawing that yields viable cultures capable of multiple passages and differentiation. Even biopsy specimens can be cryopreserved and later used to generate new GI epithelium-only organoids, which is crucial for biobanking patient material. However, the biopsy specimens are cut into tissue chunks, and cryopreserved via slow freezing in ready-made, DMSO-based cryomedia, so, although very useful, it’s not a breakthrough for cryobiology. Same for the intestinal organoids which are first centrifuged and then treated like normal cells would be.
Another study developed a protocol for mouse colon organoids, supported by theoretical modeling of heat and mass transfer. That study preserved the entire organoid on a Matrigel AND (!) transplanted the thawed organoids into mouse colon to confirm regenerative capacity, which is a bigger step for cryobiology.
Brain organoids ✅
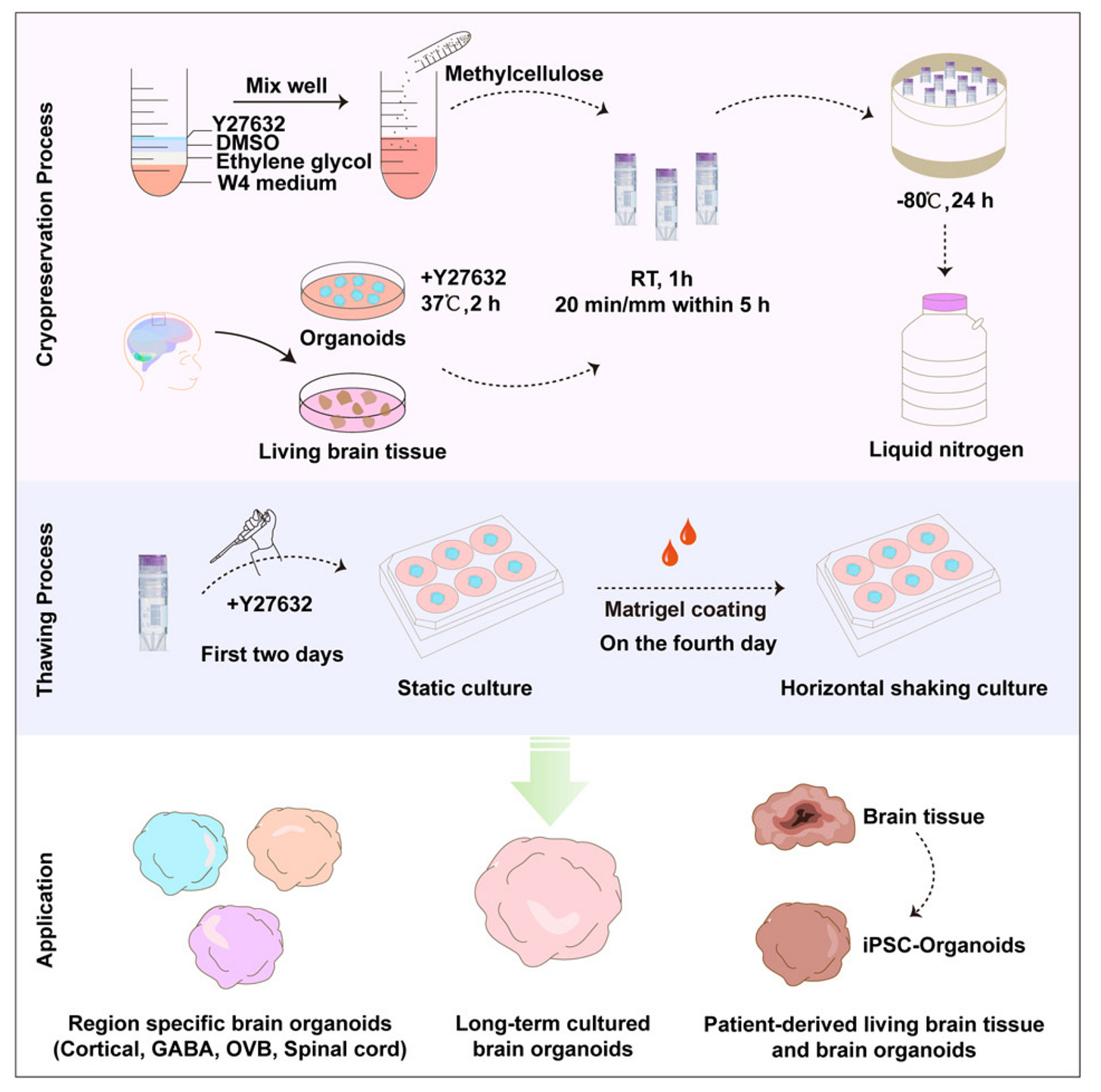
You’d be surprised to know that there is a lot of progress in brain organoid cryopreservation! In one of my favorite papers in cryo ever, Xue et al. (2024) preserved large, multilayered human brain organoids without destroying their architecture or neural identity. They generated cortical organoids from H9 human embryonic stem cells and allowed them to mature to day 21–28, at which point they contain ventricular-zone-like radial structures, proliferative Sox2⁺ neural progenitors, differentiated MAP2⁺ neurons, and early network activity. Crucially, the authors cryopreserved intact whole organoids, not dissociated clusters or centrifuged pellets, selecting spheres of comparable size before freezing to avoid size-dependent variability. Organoids were immersed directly into cryopreservation medium composed of methylcellulose, 10% ethylene glycol, 10% DMSO, and the ROCK inhibitor Y-27632 (they called this cocktail “MEDY”) and then frozen using a controlled cooling protocol. No pelleting step was used, and no mechanical disruption occurred before or after freezing; the entire structure was thawed intact, transferred back into culture, and evaluated as a single unit. The strength of the study lies in how comprehensively they quantified post-thaw recovery. First, morphological integrity was strikingly well preserved: organoids maintained their ventricular-zone-like architecture and layered cortical organization, something the field had previously failed to achieve. The MEDY organoids did not fragment, swell abnormally, or collapse into debris (common outcome under DMSO-only conditions, which produced large amounts of cell debris and even complete dissociation in the sucrose groups). Quantitatively, MEDY-treated organoids showed high retention of Sox2⁺ progenitors and MAP2⁺ neurons, each >1.5-fold higher than in DMSO-only controls, and apoptotic cells were minimal, in stark contrast to trehalose-, sucrose-, glucose-, and proline-supplemented media, which produced extensive cell death. Functionally, the thawed organoids recovered robust neurite extension when embedded in Matrigel: axonal projections in the methylcellulose–ethylene glycol conditions (the core of the MEDY formulation) were significantly longer than those preserved with DMSO alone, demonstrating that not only cell survival but neuronal outgrowth capacity was maintained. The MEDY protocol scaled beyond a single organoid type, successfully preserving dorsal forebrain, ventral forebrain, optic vesicle, spinal cord, and even epilepsy patient-derived organoids without architectural collapse or loss of regional identity. The next step for future studies would be to functionally test them.
Heart organoids ✅
Do you recall nanowarming with iron oxide particles from my last post? It turns out this technique enabled cardiac organoid cryopreservation! The development of a heart organoid vitrification workflow coupled to Fe₃O₄-based magnetic nanowarming, enabled rapid, spatially uniform rewarming of vitrified cardiac organoids. In this system, post-thaw heart organoids show significantly improved survival, beating rate, contractility, histological integrity, and multi-electrode array readouts compared with conventional slow-warming approaches, suggesting that nano-rewarming can mitigate thermal stress and ice-related damage in larger 3D cardiac constructs.
Lung, Liver and pancreatic organoids 🫤
I have not found papers that cover whole liver organoid cryopreseration, but there has been a paper that successfully cryopreserved and later cultured, smaller chunks of the liver organoid. For lungs and pancreatic islets, I only found papers that describe cryopreserving cells, and growing an organoid from thawed cells.
Kidney Organoids ✅
Human kidney organoids derived from iPSCs or primary tissue can now be reliably cryopreserved as intact whole structures. A 2024 vitrification protocol achieved ~91% post-thaw viability, preserved nephron segments (podocytes and tubules), and retained regenerative capacity against toxins like cisplatin comparable to fresh controls.
Did I miss any organoids you found interesting in terms of cryo? Let me know in the comments:
What is the next frontier?
The first frontier is functionally validated preservation. Many protocols define “success” as post-thaw morphology plus a viability stain. That may be adequate for routine passaging, but it is not enough for organoids used as quasi-clinical surrogates. The second frontier is controlled CPA delivery and removal. The main technical bottleneck is still how to load enough cryoprotectant to avoid ice, without inflicting lethal osmotic stress or toxicity. The third frontier is volumetric nanowarming at organoid scale. Nanowarming has already demonstrated major advantages over convective rewarming in perfused rat hearts and large CPA volumes: faster, more uniform heating and dramatically reduced risk of devitrification.Finally, there is a conceptual frontier: standardization and biobanking. Right now, every lab has its own slightly modified “house protocol.” As organoids move toward clinical and regulatory environments, we will need harmonized, validated cryopreservation workflows that define acceptable viability thresholds, functional readouts, and molecular fidelity benchmarks.
How to get to the next frontier of organoid cryopreservation?
The review paper by Han et al (2024) from Dr Yi Xu’s lab where I have the honor of practicing at, outlines the roadmap for organoid cryopreservation that extends beyond saying “better CPAs, less thermal gradients”. That paper makes it clear that the future of organoid cryopreservation depends on technologies that solve the physical constraints of 3D tissues. Microfluidics is one of the most transformative directions: instead of dunking organoids into high-dose vitrification solutions, microfluidic platforms allow precise, model-based CPA loading, using controlled flow, stepwise concentration ramps, and real-time osmotic tuning to avoid the catastrophic swelling and shrinkage that occur in bulk methods. These chips also let researchers measure permeability parameters directly in organoids, giving the field its first chance to design CPA schedules based on kinetics rather than guesswork. On the vitrification side, the review emphasizes “minimal-volume” formats such as Cryomesh, Cryotop, and hollow fibers, all of which achieve faster cooling and warming by physically reducing the thermal mass around each organoid. A Cryomesh, for example, spreads organoids across a thermally conductive lattice that wicks away heat during plunging, while ultrathin films on Cryotop devices allow vitrification using only microliters of solution meaning higher cooling/warming rates without raising CPA concentration.
For short-term storage, the paper highlights technologies that avoid freezing entirely. Oil-sealed supercooling keeps organoids in a metastable, ice-free state by physically isolating the medium from air, preventing heterogeneous nucleation and allowing storage at −4 to −20 °C without vitrification. Isochoric supercooling takes the idea further: by sealing samples in a rigid, constant-volume chamber, any attempt at ice formation immediately raises pressure, suppressing further crystallization. This allows low-CPA or CPA-free preservation with surprisingly high functional recovery in tissues like islets and engineered cardiac constructs.
Have you heard of any of these technologies? Let me know in the comments!
Why does organoid cryopreservation matter?
If you are like me your initial reaction to this substack would be “okay, okay, cool, but let’s move to organs, or anything that brings us closer to the whole-organism cryo”. However, just like it was shown in the Han et al (2024) review, organoids can be used as a testing ground not only for drug discovery, but also for novel cryotech, giving more reliable and scalable results than cell cultures.
For drug discovery and toxicology, cryopreserved organoid banks would allow standardized, repeatable assays across batches, sites, and years. For personalized medicine, the ability to freeze and share patient-derived organoids would underpin multicenter trials where different interventions are tested on genetically matched tissue. For regenerative medicine, cryopreservation is the only way to decouple the timing of tissue manufacture from the timing of clinical need, especially for pancreatic, hepatic, and cardiac constructs destined for transplantation. So if you are not convinced that the cool cryotech will storm from organoid cryo research, at least be certain that there is a lot of capital that can be activated for organoid banking.
There’s also a deeper continuity here. The same problems we are solving for organoids (CPA toxicity, ice avoidance, thermal stress, nanowarming) are exactly the ones limiting whole-organ cryopreservation and long-term organ banking. Techniques developed for organoids will feed back into organ work, and vice versa. In that sense, every incremental improvement in organoid cryopreservation is a step toward a future where storing complex human tissues, on demand and at scale, is normal rather than aspirational.