Three Most Impactful Recent Cryo Publications
Are you a VC doing cryo start-up due dilligence? A researcher trying to keep up? Did you sign up for cryopreservation and you're estimating your revival science-based prognosis? This post is for you
I feel like the world of cryopreservation is getting more and more buzz lately, but the state of the art still remains the 2023 rat kidney successful cryopreservation, validated by transplantation. Has the needle moved forward at all recently? I analyzed the google scholar profiles of the most impactful PIs in cryo, and then did a more general key word search in the attempt to answer this question. Here is my subjective take on the three most impactful recent publications in cryobiology.
1) Scaling Vitrification to Larger Organs
Gangwar, L. et al. (2025). “Physical vitrification and nanowarming at liter-scale CPA volumes: toward organ cryopreservation”
Nature Communications: https://www.nature.com/articles/s41467-025-63483-2
This paper will likely be cited as the 2025 cryopreservation publication.
What they did:
The authors developed a protocol to take multi-liter volumes of cryoprotectants (CPAs), big enough for human organs, and vitrify them without ice. They combined controlled cooling with nanowarming (using iron oxide nanoparticles activated by magnetic fields) for fast, uniform reheating. They also vitrified porcine livers without ice formation, but did not rewarm them for this study.
Why it matters:
So although this is just a proof of concept for bags of CPAs, rather than actual organs, this study addresses two of the hardest problems in large-scale cryopreservation:
Avoiding heterogeneous ice formation during cooling, and
Preventing cracks and devitrification during warming.
This work demonstrated avoidance of the former in pig livers, and latter with the liter volume CPAs.
Impact:
For the first time, organ-sized vitrification seems engineerable. This is the clearest line of sight for organ cryobanking there’s ever been. And although the largest successfully rewarmed organ, validated by transplantation, remains the rat kidney (also by Bischof group), this paper sends the message “we are going after human organ cryobanking”, which is a big step for academia-based cryo.
Other fun facts from the paper:
I really liked the figures where they approximate the necessary cooling and warming rates for selected organs, as a function of their adult-size-volume. It shows that the minimum cooling rate decreases with volume, which is not intuitive. Let’s talk a little bit more science. What the model is really capturing is geometry and thermal diffusion. Larger organs have a larger characteristic length scale (Lc), which fundamentally changes how heat is transported to the center. The center becomes the bottleneck. Once the surface vitrifies and thermal gradients establish, the minimum rate required to avoid ice nucleation at the center is governed by bulk heat transport, not just surface kinetics.
Panel (b) is even more interesting to me.
The blue curve shows how the minimum cooling rate collapses rapidly as Lc increases. For small characteristic lengths (sub-centimeter), the required cooling rates are extremely high. But as you move toward organ-scale dimensions, the required rate approaches very low values. At the same time, the red curve shows the tradeoff: as Lc increases, the minimum vitrifiable CPA concentration increases. So while the cooling constraint relaxes, the chemical toxicity and vascular damage due to viscous liquid perfusion constraints tighten. That tradeoff is the real engineering battlefield.
Did you think I’d stop with the science talk so soon? You must not read enough of my posts;)
The actual fun is with the figure about nanowarming. First: nanowarming is fundamentally different from conductive warming.
In traditional warming, heat diffuses from the surface inward: Bigger organ → longer diffusion path → slower center warming. That produces smooth scaling behavior. Nanowarming does not rely primarily on surface heat flux. Instead, iron oxide nanoparticles distributed through the vasculature absorb energy from an alternating magnetic field and generate heat volumetrically. The heating rate depends on:
Nanoparticle concentration
Magnetic field strength and frequency
Distribution uniformity
Vascular volume fraction
Local perfusion accessibility
Thermal conductivity of tissue + CPA
That already tells us it won’t scale cleanly with organ volume.
This paper shows that nanowarming rate correlates with vascular fraction, not volume. Organs with higher vascular density (lung, eye) show higher predicted nanowarming rates. Organs with lower vascular fraction (ovary, testis) show lower rates even if their volumes are comparable or larger. So the dominant scaling variable here is not size, but how well you can perfuse nanoparticles into the tissue. You cannot assume constant nanoparticle concentration across organs. Perfusion resistance, capillary density, endothelial permeability, and vascular geometry differ significantly.
On the note of nanowarming, another 2025 paper that deserves a shoutout is “Scalable Purification of Iron Oxide Nanoparticles for Organ Cryopreservation and Transplantation” also from the Bischof group. I’m placing it as a sub-mention, because although it didn’t show us a new proof of concept, it’s crucial for the field because it gives an exact recipe for the nanoparticles that I described above. The group could have as well formed a company around it and cash the ready-made product. I highly recommend everyone in the field to read it and understand it, for higher-order ideation on the new cryo solutions.
2) Functional Revival of the Mouse Hippocampus Cells After Whole-Brain Vitrification
Preprint: “Functional recovery of the adult murine hippocampus after cryopreservation by vitrification”
bioRxiv, https://www.biorxiv.org/content/10.1101/2025.01.22.634384v3
This preprint matters, because instead of just survival (the days of which as a valid cryo endpoint for cells are largely over) it shows function.
What they showed:
The part of the paper that is the most exciting for me shows that they cryopreserved the entire mouse brain, and although the whole organ was clearly damaged, the hippocampus, or at least the cells extracted from it, show electrochemical functionality. The whole brain was clearly stressed. There were perfusion challenges, dehydration issues, variability in recovery rates, and a non-trivial failure fraction.
Why it matters:
This is not a “perfect organ recovery” story, but it’s the largest, partially successful, published attempt at brain cryopreservation.
3) Increased understanding of organ cracking - and how it can be prevented
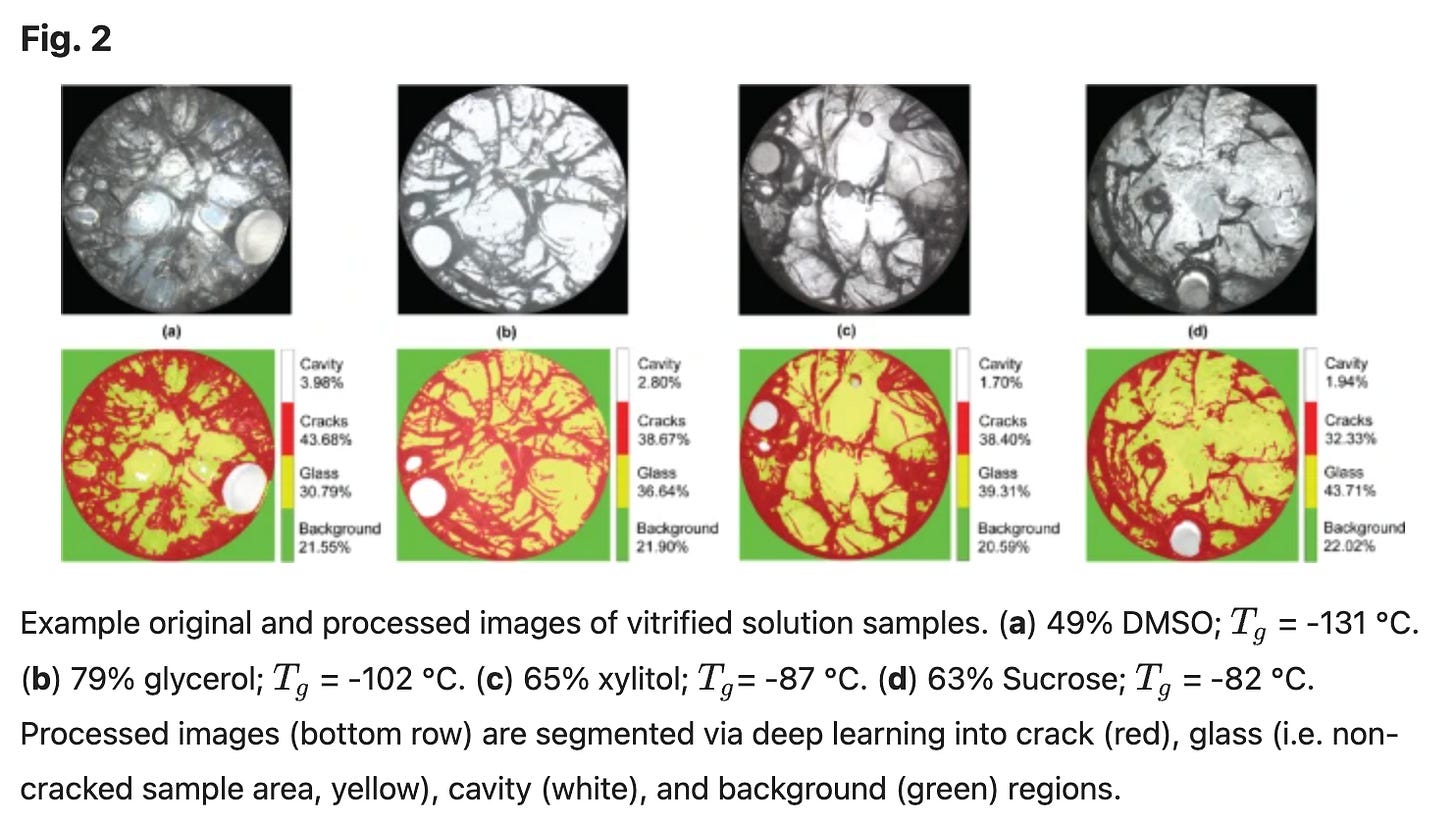
Paper: Higher Glass Transition Temperatures Reduce Thermal Stress Cracking in Aqueous Solutions Relevant to Cryopreservation
Scientific Reports - https://www.nature.com/articles/s41598-025-13295-7
Problem:
One of the major problems in organ cryopreservation is that they tend to crack during warming. Cooling and warming generate temperature gradients, which produce tensile stress in brittle aqueous glasses. Moreover, almost all CPA cocktails have a narrow glass transition temperature (Tg) band (≈ −120 to −130 °C), and Tg itself has rarely been treated as a controllable mechanical variable.
This paper asks what if fracture risk is fundamentally tied to Tg?
What they did:
The authors vitrified and rewarmed separate DMSO, glycerol, xylitol, and sucrose solutions directly imaging how much cracking occurred. Importantly, they differ in their Tg by more than 50 °C (concentration-dependent). They then built a mechanical model to calculate how much tensile stress develops during cooling for solutions with different Tg values. Both the experiments and the simulations showed the same result: solutions with higher glass transition temperatures cracked less and experienced lower peak tensile stress during cooling. In the simulations, lowering Tg increased the maximum stress by roughly fourfold across the range studied
Why it matters
This paper suggests that the dominant CPA chemistries may be thermomechanically suboptimal. Although they allow us to vitrify large samples, their Tg makes them prone to stress accumulation. This work reframes Tg as a design variable in next-generation CPAs.
Looking Forward
We’re still early in the timeline from proof-of-principle to clinical utility for organ cryobanking. Let alone revival of the whole organism. Major hurdles remain:
Full organ post-thaw functionality (especially vascular integrity)
Standardized CPA formulations with minimal toxicity
Integrated warming methods compatible with clinical workflows
But the research published in 2025 shows those hurdles are ripe for engineering and scientific attack, and that practical cryopreservation is transitioning from science fiction to scientific reality.
Did I miss any of the papers you found impactful? Are you looking for answers for specific cryo questions? Let me know in the comments!






The most important contribution of this paper is demonstrated rapid warming on a human organ scale. The cooling part, vitrifying multi-liter volumes of M22, is actually easy because M22 was originally designed and demonstrated for that purpose more than 20 years ago. By coincidence, I first showed that two liter volumes of M22 could be vitrified without ice formation or cracking at the University of Minnesota in 2005 during the Society for Cryobiology meeting there. ( “Toward large organ vitrification: extremely low critical cooling and warming rates of M22 vitrification solution,” Cryobiology 51, 362 (2005)). One of the slides from that talk, a photo of a vitrified 2 liter volume of M22, has been on the web for 20 years.
https://web.archive.org/web/20051220043911/http://www.alcor.org/sciencefaq.htm
Although not in refereed scientific literature, Alcor was the first to demonstrate a decade ago by CT scanning that large organs (a human brain) could be vitrified without ice formation using M22. The U of M work is certainly great and a major milestone, but the cooling problem hasn't really been a problem with M22 provided that adequate concentrations reach all parts of organs. That was what the solution was designed for.
I do not quite understand why you say that "It shows that the minimum cooling rate decreases with volume, which is not intuitive" when discussing Fig. 8 of the first paper. Perhaps it is because the graph should actually say what the figure title says "achievable cooling rates". It is quite intuitive that the larger the object, the longer it takes to cool it, most people would say so when asked if it a large pot of hot soup would cool faster than a small bowl.